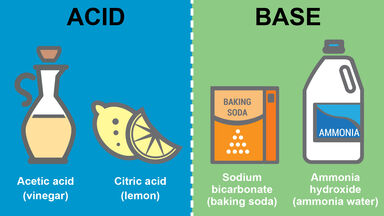
Acid Definition
ăsĭd
acids
noun
acids
Any of a class of substances whose aqueous solutions are characterized by a sour taste, the ability to turn blue litmus red, and the ability to react with bases and certain metals to form salts.
American Heritage
A sour substance.
Use vinegar or another acid in the marinade to tenderize the meat before grilling.
Webster's New World
LSD.
Webster's New World
Any compound that can react with a base to form a salt, the hydrogen of the acid being replaced by a positive ion; in modern theory, any substance that produces a positive ion and accepts electrons from a base to form covalent bonds: in water solution an acid tastes sour, turns blue litmus paper red, and, in the dissociation theory, produces free hydrogen ions.
Webster's New World
A substance having a sour taste.
American Heritage
adjective
Of, relating to, or containing an acid.
American Heritage
Sharp and biting to the taste; sour; tart.
Webster's New World
Sharp or sarcastic in temperament or speech.
Webster's New World
That is, or has the properties of, an acid.
Webster's New World
Having too heavy a concentration of acid.
Webster's New World
Synonyms:
abbreviation
(databases) Atomicity, consistency, isolation, and durability: a set of properties that guarantee database transactions are processed reliably.
Wiktionary
Other Word Forms of Acid
Noun
Singular:
acidPlural:
acidsOrigin of Acid
-
From Latin acidus sour from acēre to be sour ak- in Indo-European roots
From American Heritage Dictionary of the English Language, 5th Edition
Find Similar Words
Find similar words to acid using the buttons below.