Vaporization Definition
noun
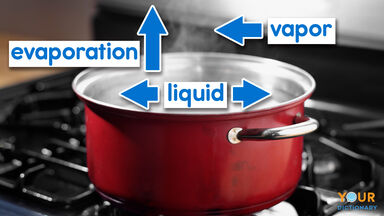
A conversion of a solid or a liquid into a gas.
Wiktionary
A destruction of something by turning it into vapor.
Wiktionary
Synonyms:
Other Word Forms of Vaporization
Noun
Singular:
vaporizationPlural:
vaporizationsOrigin of Vaporization
-
vapor + -ization
From Wiktionary
Vaporization Is Also Mentioned In
Find Similar Words
Find similar words to vaporization using the buttons below.