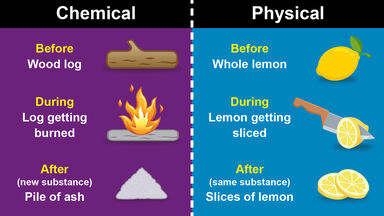
Substance Definition
- with regard to essential elements
- actually; really
Other Word Forms of Substance
Noun
Idioms, Phrasal Verbs Related to Substance
- in substance
Origin of Substance
-
Middle English from Old French from Latin substantia from substāns substant- present participle of substāre to be present sub- sub- stāre to stand stā- in Indo-European roots
From American Heritage Dictionary of the English Language, 5th Edition
-
From Old French substance, from Latin substantia (“substance, essence"), from substāns, present active participle of substō (“exist; literally, stand under"), from sub + stō (“stand").
From Wiktionary
Substance Is Also Mentioned In
Find Similar Words
Find similar words to substance using the buttons below.