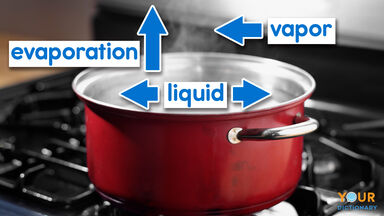
Evaporation Definition
ĭ-văpə-rāshən
noun
(archaic) That which is evaporated; vapor.
Wiktionary
Synonyms:
- vaporisation
- vapour
- vapor
- vaporization
- drying-up
- desiccation
- dehydration
- escape
- evanescence
- vanishing into thin air
- gasification
- boiling away
- steaming away
- vaporescence
- vanishing
Other Word Forms of Evaporation
Noun
Singular:
evaporationPlural:
evaporationsOrigin of Evaporation
-
From Old French évaporation, from Latin evaporatio
From Wiktionary
Evaporation Is Also Mentioned In
Find Similar Words
Find similar words to evaporation using the buttons below.