Dissociation Definition
dĭ-sōsē-āshən, -shē-
noun
The act of dissociating or the condition of having been dissociated.
American Heritage Medicine
A dissociating or being dissociated; separation.
Webster's New World
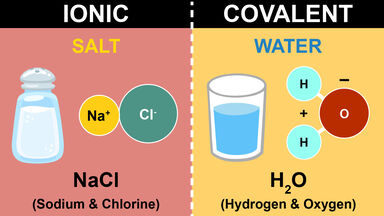
The breaking up of a compound into simpler components, as with heat or a solvent, frequently in a reversible manner so that the components may recombine.
Webster's New World
A split in the conscious process in which a group of mental activities breaks away from the main stream of consciousness and functions as a separate unit, as if belonging to another person.
Webster's New World
The abnormal separation of related ideas, thoughts, or emotions.
Webster's New World
Antonyms:
Other Word Forms of Dissociation
Noun
Singular:
dissociationPlural:
dissociationsFind Similar Words
Find similar words to dissociation using the buttons below.