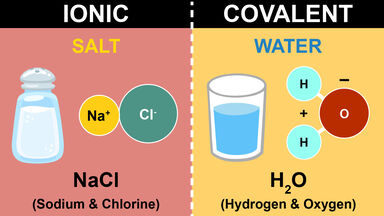
Ion Definition
An atom or group of atoms bearing an electrical charge such as the sodium and chlorine atoms in a salt solution.
Other Word Forms of Ion
Noun
Origin of Ion
-
Greek ion something that goes neuter present participle of ienai to go ei- in Indo-European roots
From American Heritage Dictionary of the English Language, 5th Edition
From Ancient Greek ἰόν (ion, “going”), neuter present participle of εἶμι (eimi, “I go”).
From Wiktionary
Middle English from Old French from Latin -iō -iōn- n. suff.
From American Heritage Dictionary of the English Language, 5th Edition
From Ancient Greek Ἴων (Iōn).
From Wiktionary
Find Similar Words
Find similar words to ion using the buttons below.