Cathode Definition
kăthōd
cathodes
noun
cathodes
In an electron tube, the negatively charged electron emitter.
Webster's New World
In an electroplating cell, the negatively charged electrode, from which current flows.
Webster's New World
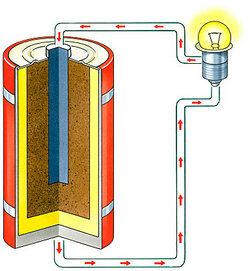
In a battery that is a source of electric current, as a dry cell or storage battery, the positive electrode which receives the electrons from the external circuit.
Webster's New World
(electronics) The electrode from which electrons are emitted into a vacuum tube or gas-filled tube.
Wiktionary
Antonyms:
Other Word Forms of Cathode
Noun
Singular:
cathodePlural:
cathodesOrigin of Cathode
Greek kathodos descent kat-, kata- cata- hodos way, path
From American Heritage Dictionary of the English Language, 5th Edition
From Ancient Greek κάθοδος (kathodos, “descent, way down”).
From Wiktionary
Related Articles
Find Similar Words
Find similar words to cathode using the buttons below.