Anode Definition
ănōd
anodes
noun
anodes
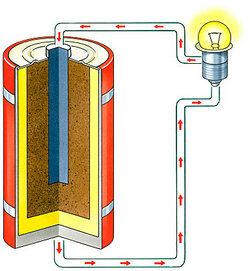
A positively charged electrode, as of an electrolytic cell, storage battery, or electron tube.
American Heritage
In an electroplating cell, the positively charged electrode, toward which current flows.
Webster's New World
The negatively charged terminal of a primary cell or of a storage battery that is supplying current.
American Heritage
In an electron tube, the principal electrode for collecting electrons, operated at a positive potential with respect to the cathode.
Webster's New World
In a battery that is a source of electric current, as a dry cell or storage battery, the negative electrode from which the electrons are released to the external circuit.
Webster's New World
Antonyms:
Other Word Forms of Anode
Noun
Singular:
anodePlural:
anodesOrigin of Anode
Greek anodos a way up ana- ana- hodos way
From American Heritage Dictionary of the English Language, 5th Edition
From Ancient Greek ἄνοδος (anodos, “way up”).
From Wiktionary
Related Articles
Find Similar Words
Find similar words to anode using the buttons below.