Saturated-solution Definition
noun
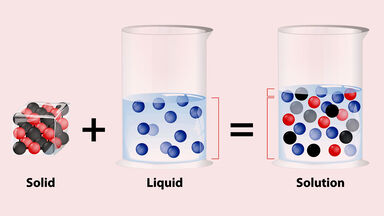
A solution in which the solvent can dissolve no more of a specific solute at a particular temperature.
Wiktionary
Other Word Forms of Saturated-solution
Noun
Singular:
saturated-solutionPlural:
saturated solutionsRelated Articles
Saturated-solution Is Also Mentioned In
Find Similar Words
Find similar words to saturated-solution using the buttons below.