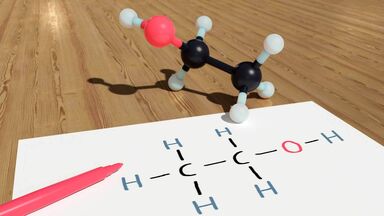
Hydrogen Bond Definition
noun
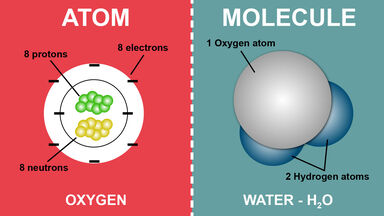
A weak chemical bond formed through a hydrogen atom, esp. if bridging two similar electronegative atoms or groups from two different molecules.
Webster's New World
verb
(chemistry) To bond to another species by means of hydrogen bonds. E.g. Water hydrogen bonds with itself.
Wiktionary
Hydrogen Bond Is Also Mentioned In
Find Similar Words
Find similar words to hydrogen bond using the buttons below.