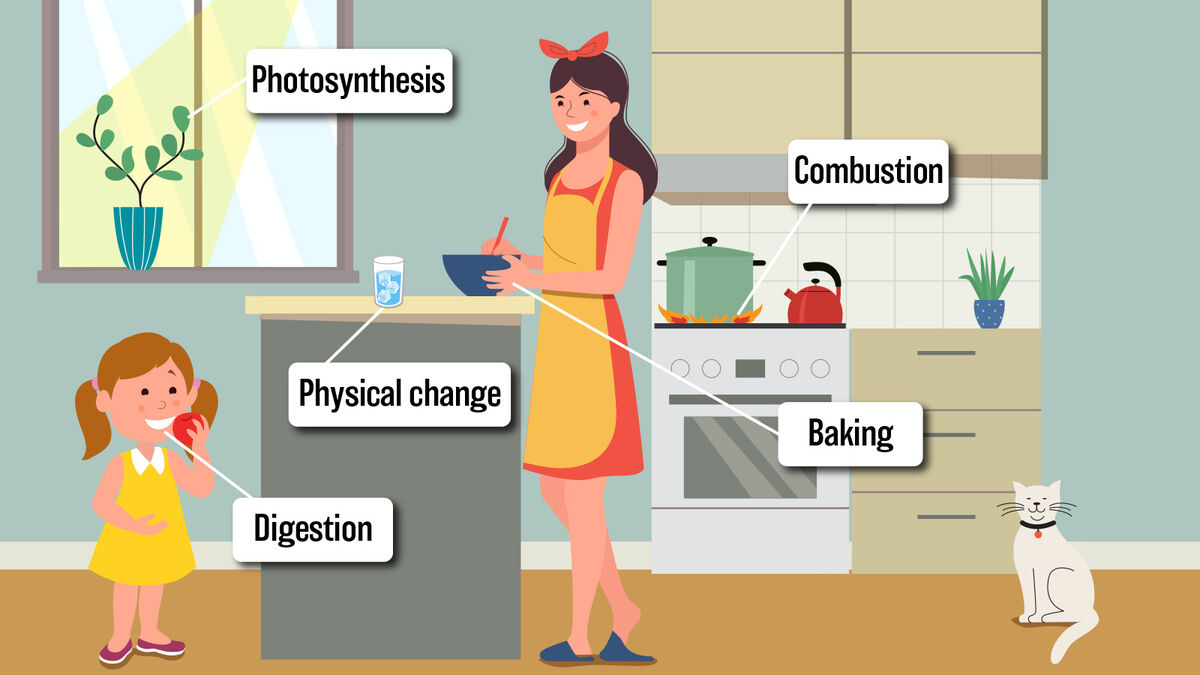
Chemical changes might bring to mind fireworks or explosions. However, some examples of chemical changes are subtle. Explore examples of chemical changes in cooking, nature, and your home. Get an understanding of the difference between physical and chemical changes.
Examples of Chemical Changes
Not all reactions are chemical. However, if a reaction makes a new substance, then you are looking at a chemical change. For example, those fireworks covering the sky on the fourth of July are the result of a chemical change. Photosynthesis in plants is also an example of a chemical reaction. In this case, sunlight is changed into food for the plant. Typically, chemical reactions have a few tells, including:
- Color changes
- Releasing or absorbing of energy
- Odor changes
- Gas production
Chemical changes happen all around you, even in your own home. Explore some fun examples in cooking, nature, and your home.
Examples of Chemical Changes in Cooking
When you mix the flour and sugar to make a cake, you might not think it's a chemical reaction at play but it is. Baking a cake is a classic example where a new substance is created from mixing flour, sugar, vanilla, cocoa, and heat. Here are a few other cooking examples.
Rising Bread
Bread rising is exciting, right? Not so much! However, not all chemical changes are exciting. While not thrilling, the yeast causing the bread to rise is a chemical change. Why? Well, yeast breaks the starches in the bread dough down into sugars, and those sugars metabolize into carbon dioxide and ethanol. The carbon dioxide makes the bread rise.
Caramelization
Do you like caramelized onions? Either way, it’s a great example of a chemical change. In caramelization, the sugar is broken down to create a specific aroma and taste.
Maillard Browning
Maillard browning is a strange name, but it's how you get that delectable brown crust on your bread and barbecued meat. Maillard browning, named for the chemist Louis-Camille Maillard, is a series of chemical reactions starting with amino acids and sugar.
Natural Examples of Chemical Changes
Cooking isn’t the only place you see chemical changes. From the way that your body digests food to a match burning when you strike it, chemical reactions can be found everywhere. Dive into a few different examples in nature and your home.
Leaves Changing Color
Those beautiful colors of fall aren’t happening by chance. They are thanks to chemical changes in the leaves. Typically, leaves are green due to chlorophyll used for photosynthesis. However, before trees take their winter nap, chlorophyll in the leaves breaks down. As the chlorophyll breaks down, the carotene and anthocyanins naturally in leaves all summer long take center stage creating those beautiful red and yellow colors.
Souring Food
One of the key characteristics a chemical change has taken place is odor, which is why people might smell foods to see if they went bad. For example, milk goes bad because of fermentation. This is where the lactose is transformed into lactic acid by bacteria surviving pasteurization.
Combustion
When something undergoes a chemical reaction, it becomes something else. For example, when wood burns, it becomes ash. The reason wood burns is due to combustion.
Mixing Baking Soda and Vinegar
If you mix an acid, such as vinegar, with a base, like baking soda, you’ll get a chemical reaction. In this case, it starts to bubble. This is a great way to create a homemade volcano.
Car Rusting
You never want your car to rust. However, no matter where you live, it’s inevitable. Rust is the chemical change taking place when iron is exposed to metal. Unless your car is made of plastic or in a bubble, it will find a way to rust.
Chemical Change vs Physical Change
An area of confusion in chemistry can be the difference between chemical and physical changes. While a chemical change is when something becomes a new substance, a physical change is when a substance changes form but stays the same. For example, when water is frozen, it changes form but is still water. When water is evaporated into the air, it changes form but is still water. If you shred a block of cheese, the cheese is still cheese, but it changed shape.
Fun Chemical Changes
Chemistry can be fun. If you’ve ever made a homemade volcano, you know this can be exciting. However, not all changes are chemical changes. Chemical changes only happen when a new substance is formed. With a solid grasp on chemical changes, check out biotic factors and abiotic factors.