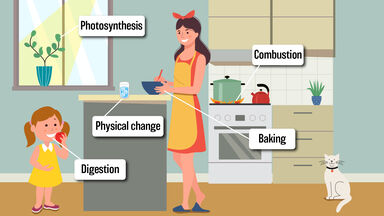
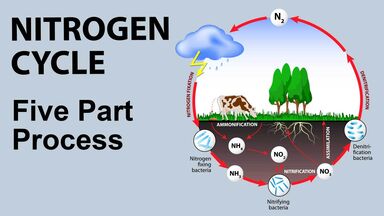
Chlorophyll Definition
klôrə-fĭl
noun
The green pigment found in the chloroplasts of plant cells: it occurs in five forms, esp. (chlorophyll a), C55H72MgN4O5, and (chlorophyll b), C55H70MgN4O6: it is essential to the photosynthetic process and is used as a coloring agent, in topical medicines, etc.
Webster's New World
Any of a group of green pigments that are found in the chloroplasts of plants and in other photosynthetic organisms such as cyanobacteria.
Wiktionary
Synonyms:
- chlorophyl
Other Word Forms of Chlorophyll
Noun
Singular:
chlorophyllPlural:
chlorophyllsOrigin of Chlorophyll
-
From French chlorophylle.
From Wiktionary
Related Articles
Find Similar Words
Find similar words to chlorophyll using the buttons below.