Calcium Hydroxide Definition
noun
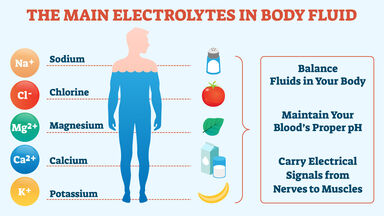
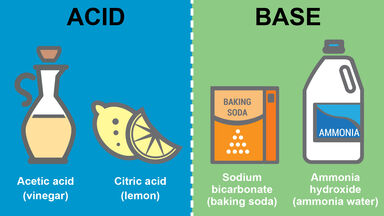
Slaked lime, Ca(OH)2, a white, crystalline compound prepared by the action of water on calcium oxide, used in making plaster, mortar, alkalies, bleaching powder, etc.
Webster's New World
Synonyms:
- calcium hydrate
- hydrated lime
- slaked-lime
- lime hydrate
- lime
- caustic lime
Calcium Hydroxide Is Also Mentioned In
Find Similar Words
Find similar words to calcium hydroxide using the buttons below.