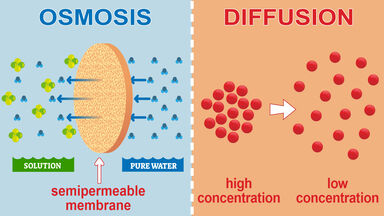
Much more important is the effect of the alteration in the amount of crystalloids in the tissues and blood and therefore of the alteration in the osmotic pressure between these.
Thus differences in osmotic pressure may be much more powerful in producing oedema than mere differences in blood pressure.
Now differences in the amount of crystalloids cause alteration in osmotic pressure while the proteid content affects it but little; and of the crystalloids the chlorides appear to be those most liable to variation.
Some other observers, however, have not got such good results with a chloride-free diet, and Marishler, Scheel, Limbecx, Dreser and others, dispute Widal's hypothesis of a retention of chlorides as being the cause of oedema, in the case of renal dropsy at all events; they assert that the chlorides are held back in order to keep the osmotic pressure of the fluid, which they assume to have been effused, equal to that of the blood and tissues.
Thus, while increased pressure in the blood or lymph vessels may be one factor, and increased permeability of the capillary endothelium another, increased osmotic pressure in the tissues and lymph is probably the most important in the production of dropsy.