Nitrogen Definition
nītrə-jən
noun
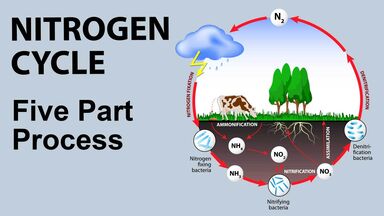
A colorless, tasteless, odorless, gaseous chemical element forming nearly four fifths of the atmosphere: it is a component of all proteins and nucleic acids: symbol, N; at. no. 7
Webster's New World
(countable) A specific nitrogen within a chemical formula, or a specific isotope of nitrogen.
The two nitrogens are located next to one another on the ring.
Wiktionary
Synonyms:
- atomic number 7
- n
Other Word Forms of Nitrogen
Noun
Singular:
nitrogenPlural:
nitrogensFind Similar Words
Find similar words to nitrogen using the buttons below.