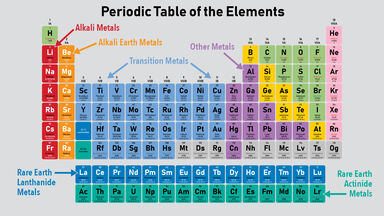
Alkali Definition
ălkə-lī
alkalies, alkalis
noun
alkalis
A carbonate or hydroxide of an alkali metal, the aqueous solution of which is bitter, slippery, caustic, and characteristically basic in reactions.
American Heritage
Any base or hydroxide, as soda, potash, etc. that is soluble in water and gives a high concentration of hydroxyl ions in solution; specif., any of the hydroxides and carbonates of the alkali metals.
Webster's New World
Any of various soluble mineral salts found in natural water and arid soils.
American Heritage
Any soluble substance, as a mineral salt or mixture of salts, that can neutralize acids, has a pH greater than 7.0, and turns litmus blue: strong alkalies are caustic.
Webster's New World
Alkali metal.
American Heritage
Synonyms:
Other Word Forms of Alkali
Noun
Singular:
alkaliPlural:
alkalies, alkalisOrigin of Alkali
-
Middle English alkaline substance from calcined plant ashes from Medieval Latin from Arabic al-qily the ashes, lye, potash al- the qily ashes (from qalā to fry, roast qly in Semitic roots)
From American Heritage Dictionary of the English Language, 5th Edition
-
French alcali, ultimately from Arabic القلي (al-qilī, “ashes of the saltwort”), from قلى (qalā, “to roast in a pan, fry”).
From Wiktionary
Find Similar Words
Find similar words to alkali using the buttons below.