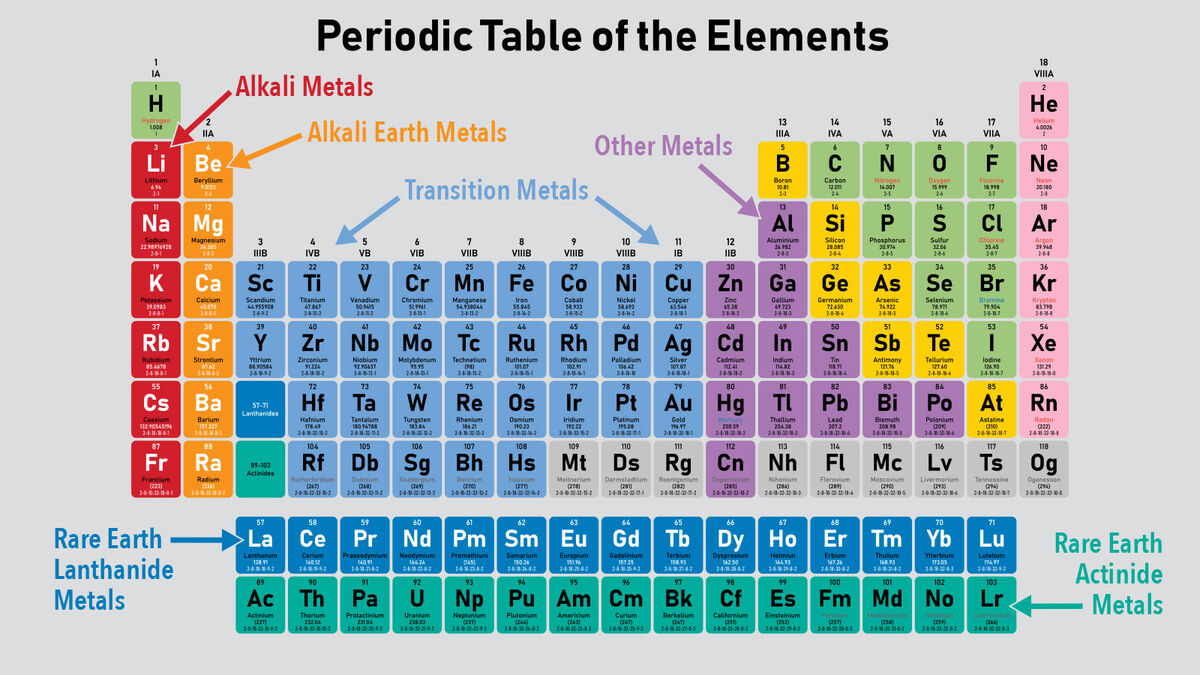
About 75% of all the known chemical elements in the world are categorized as metals. However, there are five distinct types of metals found on the Periodic Table of Elements. According to the Royal Society of Chemistry, there are 94 metals listed on the Periodic Table.
What Is a Metal?
Alkali Metals
The alkali metals can be found in the first column on the left side of the Periodic Table. They are soft metals that are highly reactive and have one electron in their outermost s sub-shell.
The six alkali metals are:
- lithium
- sodium
- potassium
- rubidium

- caesium
- francium
Alkaline Earth Metals
The alkaline earth metals are found in column 2 on the left side of the Periodic Table. They are generally harder and denser than alkali metals, have 2 electrons in their outermost s sub-shell, and each make a distinct color in their flames.
The six alkaline earth metals are:
- beryllium
- magnesium
- calcium
- strontium
- barium
- radium
Transition Metals
The transition metals are found in the center of the main body of the Periodic Table. They are sometimes called heavy metals and are denser than alkali or alkaline earth metals.
There are 38 transition metals including:
- cobalt
- copper

- gold
- iron
- mercury
- platinum
- silver
- titanium
- tungsten
- zinc
Rare Earth Metals
The rare earth metals are typically found in their own table below the main Periodic Table. However, they actually fit into the middle of the Periodic Table. There are two types of rare earth metals: lanthanides and actinides.
Lanthanide Metals
There are 15 lanthanides listed on the Periodic Table. All of these elements are so similar, it is very difficult to differentiate them.
Examples of lanthanides include:
- cerium
- promethium
- gadolinium
- dysprosium
- lutetium
Actinide Metals
There are 15 actinides listed on the Periodic Table. Most of these do not occur in nature because they are so unstable but are produced in nuclear reactors and particle accelerators.
Examples of actinides include:
- thorium
- uranium
- plutonium
- californium
- mendelevium
Other Metals
Metals listed as other metals are found toward the right side of the main body of the Periodic Table. These are sometimes called semimetals and sometimes called post-transition metals. They are typically softer than other metals and have lower melting points.
Scientists don’t agree universally which elements belong in this category, so depending on the table you’re looking at, there might be anywhere from 8-14. Examples of other metals include:
- aluminum
- bismuth

- indium
- lead
- tin
The Elements of Metal
The types of metals on the Periodic Table can be further broken down by other properties, but these are the basic types of metals. Continue exploring the Periodic Table by checking out element examples from all the types of elements.