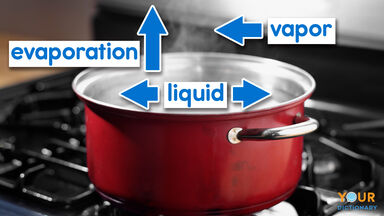
Water Vapor Definition
noun
Water in the form of a gas; steam.
Webster's New World
Water in a gaseous state, especially when diffused as a vapor in the atmosphere and at a temperature below boiling point.
American Heritage
Wiktionary
Synonyms:
- water-vapour
adjective
Attributive form of water vapor, noun.
Water-vapor production.
Wiktionary
Other Word Forms of Water Vapor
Noun
Singular:
water vaporPlural:
water-vaporsWater Vapor Is Also Mentioned In
Find Similar Words
Find similar words to water vapor using the buttons below.