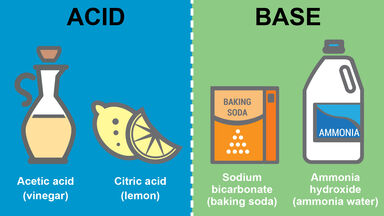
Citric Definition
sĭtrĭk
adjective
Of or relating to citric acid.
American Heritage
Of or from citrus fruits.
Webster's New World
Designating or of an acid, C6H8O7, obtained from such fruits, used in making flavoring extracts, dyes, citrates, etc.
Webster's New World
Find Similar Words
Find similar words to citric using the buttons below.