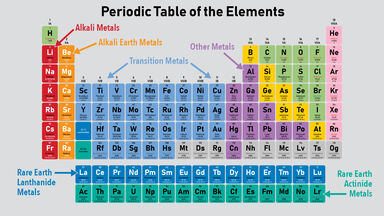
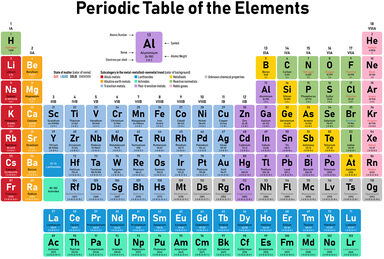
Transition Element Definition
trăn-zĭshən
noun
Any element of several groups of elements formed by adding electrons to an inner shell as the atomic number increases.
Webster's New World
Other Word Forms of Transition Element
Noun
Singular:
transition elementPlural:
transition-elementsTransition Element Is Also Mentioned In
Find Similar Words
Find similar words to transition element using the buttons below.