Calorimeter Definition
kălə-rĭmĭ-tər
calorimeters
noun
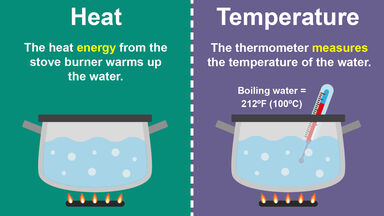
An apparatus for measuring amounts of heat, as in chemical combination, friction, etc.
Webster's New World
Other Word Forms of Calorimeter
Noun
Singular:
calorimeterPlural:
calorimetersOrigin of Calorimeter
-
Latin calor heat kelə-1 in Indo-European roots –meter
From American Heritage Dictionary of the English Language, 5th Edition
calorie- + -meter
From Wiktionary
Find Similar Words
Find similar words to calorimeter using the buttons below.