Bohr Theory Definition
noun
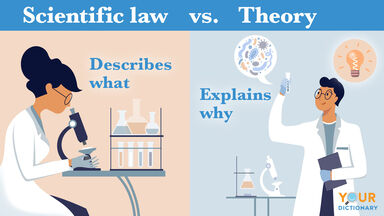
A theory, proposed by Niels Bohr in 1913, stating that electrons revolve in definite orbits around a nucleus, and that radiation is absorbed or emitted only when an electron is transferred from one orbit to another.
Webster's New World
Origin of Bohr Theory
After Niels Henrik David Bohr
From American Heritage Dictionary of the English Language, 5th Edition
Find Similar Words
Find similar words to Bohr theory using the buttons below.